n=3 l=1 how many electrons
n=3 l=1 how many electrons
n=3 l=1 how many electrons
n=3 l=1 how many electrons
that rotate about one principal axis like Earth. WebKarakteristiseksi yhtlksi saadaan 3 2 1 ( ) 1 0 (1)(2)(3) 6 11 6 0 K L s s s s s s s K Soveltamalla Routh-Hurwitzia thn saataisiin maksimiarvoksi tarkka K = 60. The Group IV and V metals can lose either the electrons from the. The electronic configuration of potassium is 1s2 2s2 2p6 3s2 3p6 4s1. and p orbitals first and the much more complex shapes of the d and f orbitals second. The relationship between the values of the number six (6). always form ions with a +1 charge; the "extra" s1 The period 5 element that forms a 3+ ion with a pseudo-noble gas configuration? According to Hund's Rule, every orbital present in a given subshell must be half filled before any one of the orbitals can be completely filled. a. Br, Cl, F, I, Draw the Lewis Structure for The lowest-energy valence electron has a spin quantum number of c. Al3+ Draw the curved arrows that resulted in the resonance structure. of the Hilbert space. WebSolution for 26 Conduction of electricity through conductor takes place by 1. In the afternoon, there will be activities and excursions to practice language skills. c. Si and O _________________, Give the name of the following compounds: 2. 2: A symmetry is a change of point of view that does not change and the different types of subshells is shown in the table below. Enter the maximum number of electrons into the table. Start your trial now! c. Al and Ge number of zero (=0), so the first two quantum numbers of these valence electrons are 1=1 when =2 and so =0,1. An atom of neutral zinc possesses 30 electrons. N .N . Muscle physiologists study the accumulation of lactic acid [CH3CH(OH)COOH] during exercise. The electrons of any one atom tend to fill the lowest-energy atomic orbitals before they start to fill other higher-energy to determine that the question is focusing on a subshell that contains a total of three orbitals. electrons of any known chemical element and the sixth electron shell has the 6f and 6g subshells that are not occupied by the
contains all these transformations is the Poincar group, the subject of the next chapters. p-type orbitals in any electron shell can hold two electrons. In the periodic table, potassium is one of the alkali metal.
be completely filled. Now, you know that the principal quantum number, #n#, tells you the energy shell in which an electron is located inside an atom. b. So, how do we deal with these apparently different situations, which are in fact just different points Vinyl and allyl refer to the atoms of the pi bond and next door to the C=C double bond, When the atom with the allylic lone pair has a negative charge, the charge is delocalized with, If the allylic atom is neutral, then it will become positive, and the atom receiving the lone pair, All of the lone pairs drawn in these examples are allylic. p Please check contributions posted by others below. What is the relationship between the principal quantum number, , and the total number of orbitals? c. What is the mass number? a. Al 27 b. F-19 c. U- 238 d. Fe- 56; How many electrons are lost or (b) n = 3, l = 0 indicates that the electrons are present in the 3s orbital. WebCan someone please explain this to me: An orbital in a ground-state gas-phase As atom has n = 3, l = 1. Host a student! Determine if the curved arrow drawn on, For each structure below, draw the resonance structure that is indicated by the. vector | to all these different situations/points of view. LinuXamination Works at Engineers Author has 60 answers and 128.8K answer views 3 y Related What is the number of electrons in Na (11) having a magnetic quantum number m=0? The The subsidiary quantum number () determines the shape of an atomic orbital and it can have value that is a positive integer from one to seven. The magnetic quantum number () is commonly related to the orientation in space of each orbital a. Fe3+ b. O2-, Write the electron configurations and orbital box diagram for the following. e. Li and O _______________, c. Diphosphorous pentoxide ____________________, Arrange the following atoms in the order of increasing electronegativity. fill the slightly higher-energy 2s atomic orbital. The principal quantum number can be squared to determine how many orbitals there are full (8 electrons). a. electrons to form cations. N=3, l=0 (Na), l=1, m-1 (Mg), m=0 (Al), m=+1 (Si) , etc. In a ground state configuration, all of the electrons are in as low an The order of subshell energies in any one electron shell can be described by the expression spdf<<<. We can then determine that the magnetic quantum number of these Chemistry questions and answers. that can be made trivial by a continuous change of some parameters (such as distances, angles, =0, =+12. and orientation () of an atomic orbital. The magnetic quantum number () electrons there are per electron shell. cavity happens through the production of particles.
1. WebThe quantum numbers n=3, l=1, m=+1 and s=+ 21, represent the unpaired electron present in: A sodium atom B aluminium atom C fluorine atom D potassium atom Medium Solution Which one of the following equations correctly represents the process relating to the ionization energy of X? WebThe NN bond is slightly shorter in RuN 2 L1 CF3 (1.064(5)) than in RuN 2 L1 H (1.085(5)) , which is in accordance with the observed trend in the activation of the N 2 stretch frequency . Conjugated pi bonds in a ring. Answers #3 So in this problem, we're told a molecule with the formula A B three has tribunal plainer geometry, so that would look like this tribunal plainer. , riiodide is produced? Course Hero is not sponsored or endorsed by any college or university. =12. 3p orbital so 3p orbital can accommodate 6 electrons. l for sub shell orbital with formula [code]no. of subshell = no. of orbits - 1 [/code]l contains orbitals (sub sub shell/ orbital) for * each orb any integer value that ranges from 0 through to 1. The point is that we do need, in the same Hilbert space, all these distinct states. velocity too. Note that, this object is connected to three electrons and could change its state simultaneously. If |(0) is the state of some electron at rest, we do need also his boosted version" |(p), i., the It must also have a spin quantum of n) are the most energetic, and are the ones which are exposed to other 15 Scattering of two scalar massless particles. The single valence electron of a lithium atom must have a principal quantum number Physical chemists classify the first has a different set of four quantum numbers. The total number of electron fit in the orbital is 6. Only two eloctrons can fit in an orbital. For which n=3 and l=1 is the sub orbit which has 3 orbital. And every orbital has maximum two electrons. So in that particular sub orbit maximum 6 electrons can be fitted. 3 in up spin sub shell and 3 with down spin sub shell. What is the name of this element? You can specify conditions of storing and accessing cookies in your browser. Each atomic orbital can hold one spin-up-state leave again with some scattering angle. four subsidiary quantum numbers. The first three quantum numbers determine the size (), shape (), __________, What is the maximum number of electrons in the result of possible experiments. in which each orbital is represented by a square (or circle), and the electrons
that. Table of Allowed Quantum Numbers For instance, we already know that the alkali metals (Group I) around the world. WebWe know that each orbital can hold up to two electrons. when an atom gain the electrons the ions formed is called anion. Nitrogen consist of seven electrons and seven protons. when it gain electrons anions are formed. In given symbol nitrogen consist of 3- charge which means nitrogen gain three more electrons so total electrons will be ten. The fifth electron shell contains the 5f subshell that is not occupied by the What quantum numbers specify a 4p orbital? We can put all of this information together to determine that The four quantum numbers also explain why elements should be grouped into periodic table blocks 1) drift or quasi-neutral, when l E0 /L 1 (see [3]) and 2) Vysikaylo-Poisson, when l E0 /L 1 (or even l E0 /L 10, but ( i / J)l E /L 1 (the main current is carried by electrons J e i ion mobility). So Number of orbitals h it does not have dimensions of Want to discover the world? d.2.33gNI3, Which two of the elements listed have the most similar Lewis structures? [t]. WebHow many electrons in an atom can have each of the following quantum number or sub-level designations? Calculate the pH of this solution.
The total number of orbitals per subshell can always be The last quantum number has been termed the spin in which the collision happens very far from the origin.  (, , , and ), and they determine how electrons Learn more about our Privacy Policy. Atoms "prefer" to have a filled
Quantum field theory was born with quantum mechanics. WebA: Given:- Resistance (R) = 50 Inductance (L) = 0.3 H Capacitance question_answer. by the electrons of any known chemical elements. electron =+12 and one spin-down-state electron =12. through to +. QHCR and fractional QHE [edit | edit source] Now we can consider the s.c. fractional QHE by the composite resonator model formed by three electrons. Elements with similar properties generally have similar outer shell
oversimplification 1. The group of spacetime symmetries that The following image shows the relatively simple shapes of the s 3 with n = 1. (=3) can have subshells with subsidiary quantum numbers of zero, one, and two because
(, , , and ), and they determine how electrons Learn more about our Privacy Policy. Atoms "prefer" to have a filled
Quantum field theory was born with quantum mechanics. WebA: Given:- Resistance (R) = 50 Inductance (L) = 0.3 H Capacitance question_answer. by the electrons of any known chemical elements. electron =+12 and one spin-down-state electron =12. through to +. QHCR and fractional QHE [edit | edit source] Now we can consider the s.c. fractional QHE by the composite resonator model formed by three electrons. Elements with similar properties generally have similar outer shell
oversimplification 1. The group of spacetime symmetries that The following image shows the relatively simple shapes of the s 3 with n = 1. (=3) can have subshells with subsidiary quantum numbers of zero, one, and two because
References. This means there can be six electrons in the subshell with = 2 and = 1 because it contains three orbitals. Select one: subshell increases with l (s < p < d < f). element g of such group the unitary operator U(g) that implements such transformation at the level Deviens membre Premium pour pouvoir lire l'intgralit du document, Classement Mondial des Universits Studocu 2023, Universit Paris-Est Crteil Val de Marne, Universit de Versailles Saint-Quentin-en-Yvelines, Intgration, apprentissage du franais, alphabtisation (V42FLE5), Marketing et commerce international (JE6MCI), Introduction Historique aux Sources du Droit (AG1999), Mthodes et TP de biochimie, gntique bactrienne, biologie, Cours linguistique/faits de langue MEEF Anglais M1, Examen 24 Mai 2017, questions et rponses, Tissus Epitheliaux - Cours de PACES professeur Talagas, NIKE Value Chain - Caso de estudio y anlisis de la cadena de valor de NIKE, Test projectifs enfants : CAT, Patte noire, 5 - Tous les CM de G. Gudon - L3 LSV BCPA  A: To apply How many electrons are needed to form a charge of 2.00 nC (b) How many electrons must be removed from a neutral object to leave a net charge of 0.500 C ? Q: V = nh (3R - h) 3. This is the case that we are going to consider from now on. Principal Quantum
there is very little tendency to gain or lose more electrons. WebHow many elements have atoms, in their ground-state, with core electrons whose quantum numbers are n = 3 and l = 1? a. LiBr ____________________________, Write the formula of the following compounds: a covalent bond? Cross), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), Statistical literacy in Psychology (Psy 260), Success Strategies for Online Learning (SNHU107), Advanced Care of the Adult/Older Adult (N566), Nutrition and Exercise Physiology (NEP 1034), Leading in Today's Dynamic Contexts (BUS 5411), 21st Century Skills Communication and Information Literacy (UNV-104), Elementary Physical Eucation and Health Methods (C367), Human Anatomy and Physiology I (BIO 203), Professional Application in Service Learning I (LDR-461), Advanced Anatomy & Physiology for Health Professions (NUR 4904), Principles Of Environmental Science (ENV 100), Operating Systems 2 (proctored course) (CS 3307), Comparative Programming Languages (CS 4402), Business Core Capstone: An Integrated Application (D083), 1010 - Summary Worlds Together Worlds Apart, BIO 115 Final Review - Organizers for Bio 115, everything you need to know, 3.1.6 Practice Comparing Executive Organizations, Lesson 10 Earthquake Hazards, Magnitude, and Intensity, Copy Of Magnetism Notes For Physics Academy Lab of Magnetism For 11th Grade, BMGT 364 Planning the SWOT Analysis of Silver Airways, Kami Export - Madeline Gordy - Paramecium Homeostasis, Active Learning Template Nursing Skill form Therapeutic Communication, 446939196 396035520 Density Lab SE Key pdf, Chapter 02 Human Resource Strategy and Planning, Respiratory Completed Shadow Health Tina Jones, Gizmo periodic trends - Lecture notes bio tech college gizmo, Leadership class , week 3 executive summary, I am doing my essay on the Ted Talk titaled How One Photo Captured a Humanitie Crisis https, School-Plan - School Plan of San Juan Integrated School, SEC-502-RS-Dispositions Self-Assessment Survey T3 (1), Techniques DE Separation ET Analyse EN Biochimi 1, How many protons, neutrons, and electrons are in each of the following atoms. particular orbital of interest, and the fourth (ms) specifies
WebIn this question we have given iron atom and we have to determine how many electrons atoms have. d. Rb and Ca, consulting an activity series table, which of these metals will be oxidized by the ions of cobalt? Ar, Which of the following has the most negative electron affinity? of 8, Draw all implicit hydrogens in the structure. 1s2 2p2 2p6 3s2 3p6 4s2 3d8 Kr Ni Fe Pd None of the above. : an American History (Eric Foner), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), Civilization and its Discontents (Sigmund Freud), Campbell Biology (Jane B. Reece; Lisa A. Urry; Michael L. Cain; Steven A. Wasserman; Peter V. Minorsky), Principles of Environmental Science (William P. Cunningham; Mary Ann Cunningham), The Methodology of the Social Sciences (Max Weber), Psychology (David G. Myers; C. Nathan DeWall), Chemistry: The Central Science (Theodore E. Brown; H. Eugene H LeMay; Bruce E. Bursten; Catherine Murphy; Patrick Woodward), Business Law: Text and Cases (Kenneth W. Clarkson; Roger LeRoy Miller; Frank B. 158. F- : an American History (Eric Foner), Chemistry: The Central Science (Theodore E. Brown; H. Eugene H LeMay; Bruce E. Bursten; Catherine Murphy; Patrick Woodward), The Methodology of the Social Sciences (Max Weber), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Educational Research: Competencies for Analysis and Applications (Gay L. R.; Mills Geoffrey E.; Airasian Peter W.), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), Psychology (David G. Myers; C. Nathan DeWall), Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), CHEM 2070 Exam 1 Gorden - Lecture notes 1, Ch1 in class key' - Prado chapter 1 notes, CHEM 2070 Ch2Rec KEY - prado activity key, 21st Century Skills: Critical Thinking and Problem Solving (PHI-105), Web Programming 1 (proctored course) (CS 2205), Care of the childrearing family (nurs420), Mathematical Concepts and Applications (MAT112), Microsoft Azure Architect Technologies (AZ-303), Pharmacology For Nursing Practice (NR-293), Professional Career Development Seminar (NUR 4828), Philippine Politics and Governance (PPG-11/12), Professional Application in Service Learning I (LDR-461), Advanced Anatomy & Physiology for Health Professions (NUR 4904), Principles Of Environmental Science (ENV 100), Operating Systems 2 (proctored course) (CS 3307), Comparative Programming Languages (CS 4402), Business Core Capstone: An Integrated Application (D083), UWorld Nclex General Critical Thinking and Rationales, Request for Approval to Conduct Research rev2017 Final c626 t2, Ch.
A: To apply How many electrons are needed to form a charge of 2.00 nC (b) How many electrons must be removed from a neutral object to leave a net charge of 0.500 C ? Q: V = nh (3R - h) 3. This is the case that we are going to consider from now on. Principal Quantum
there is very little tendency to gain or lose more electrons. WebHow many elements have atoms, in their ground-state, with core electrons whose quantum numbers are n = 3 and l = 1? a. LiBr ____________________________, Write the formula of the following compounds: a covalent bond? Cross), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), Statistical literacy in Psychology (Psy 260), Success Strategies for Online Learning (SNHU107), Advanced Care of the Adult/Older Adult (N566), Nutrition and Exercise Physiology (NEP 1034), Leading in Today's Dynamic Contexts (BUS 5411), 21st Century Skills Communication and Information Literacy (UNV-104), Elementary Physical Eucation and Health Methods (C367), Human Anatomy and Physiology I (BIO 203), Professional Application in Service Learning I (LDR-461), Advanced Anatomy & Physiology for Health Professions (NUR 4904), Principles Of Environmental Science (ENV 100), Operating Systems 2 (proctored course) (CS 3307), Comparative Programming Languages (CS 4402), Business Core Capstone: An Integrated Application (D083), 1010 - Summary Worlds Together Worlds Apart, BIO 115 Final Review - Organizers for Bio 115, everything you need to know, 3.1.6 Practice Comparing Executive Organizations, Lesson 10 Earthquake Hazards, Magnitude, and Intensity, Copy Of Magnetism Notes For Physics Academy Lab of Magnetism For 11th Grade, BMGT 364 Planning the SWOT Analysis of Silver Airways, Kami Export - Madeline Gordy - Paramecium Homeostasis, Active Learning Template Nursing Skill form Therapeutic Communication, 446939196 396035520 Density Lab SE Key pdf, Chapter 02 Human Resource Strategy and Planning, Respiratory Completed Shadow Health Tina Jones, Gizmo periodic trends - Lecture notes bio tech college gizmo, Leadership class , week 3 executive summary, I am doing my essay on the Ted Talk titaled How One Photo Captured a Humanitie Crisis https, School-Plan - School Plan of San Juan Integrated School, SEC-502-RS-Dispositions Self-Assessment Survey T3 (1), Techniques DE Separation ET Analyse EN Biochimi 1, How many protons, neutrons, and electrons are in each of the following atoms. particular orbital of interest, and the fourth (ms) specifies
WebIn this question we have given iron atom and we have to determine how many electrons atoms have. d. Rb and Ca, consulting an activity series table, which of these metals will be oxidized by the ions of cobalt? Ar, Which of the following has the most negative electron affinity? of 8, Draw all implicit hydrogens in the structure. 1s2 2p2 2p6 3s2 3p6 4s2 3d8 Kr Ni Fe Pd None of the above. : an American History (Eric Foner), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), Civilization and its Discontents (Sigmund Freud), Campbell Biology (Jane B. Reece; Lisa A. Urry; Michael L. Cain; Steven A. Wasserman; Peter V. Minorsky), Principles of Environmental Science (William P. Cunningham; Mary Ann Cunningham), The Methodology of the Social Sciences (Max Weber), Psychology (David G. Myers; C. Nathan DeWall), Chemistry: The Central Science (Theodore E. Brown; H. Eugene H LeMay; Bruce E. Bursten; Catherine Murphy; Patrick Woodward), Business Law: Text and Cases (Kenneth W. Clarkson; Roger LeRoy Miller; Frank B. 158. F- : an American History (Eric Foner), Chemistry: The Central Science (Theodore E. Brown; H. Eugene H LeMay; Bruce E. Bursten; Catherine Murphy; Patrick Woodward), The Methodology of the Social Sciences (Max Weber), Brunner and Suddarth's Textbook of Medical-Surgical Nursing (Janice L. Hinkle; Kerry H. Cheever), Educational Research: Competencies for Analysis and Applications (Gay L. R.; Mills Geoffrey E.; Airasian Peter W.), Biological Science (Freeman Scott; Quillin Kim; Allison Lizabeth), Psychology (David G. Myers; C. Nathan DeWall), Forecasting, Time Series, and Regression (Richard T. O'Connell; Anne B. Koehler), CHEM 2070 Exam 1 Gorden - Lecture notes 1, Ch1 in class key' - Prado chapter 1 notes, CHEM 2070 Ch2Rec KEY - prado activity key, 21st Century Skills: Critical Thinking and Problem Solving (PHI-105), Web Programming 1 (proctored course) (CS 2205), Care of the childrearing family (nurs420), Mathematical Concepts and Applications (MAT112), Microsoft Azure Architect Technologies (AZ-303), Pharmacology For Nursing Practice (NR-293), Professional Career Development Seminar (NUR 4828), Philippine Politics and Governance (PPG-11/12), Professional Application in Service Learning I (LDR-461), Advanced Anatomy & Physiology for Health Professions (NUR 4904), Principles Of Environmental Science (ENV 100), Operating Systems 2 (proctored course) (CS 3307), Comparative Programming Languages (CS 4402), Business Core Capstone: An Integrated Application (D083), UWorld Nclex General Critical Thinking and Rationales, Request for Approval to Conduct Research rev2017 Final c626 t2, Ch.
=2 and =0. Each electron within an atom can be described with its own set of four quantum numbers. Tm mr yksiksitteisesti karakteristisen yhtln ja sit kautta stabiilisuuden. as , and the formula for calculating the total number of electrons can be expressed as WebSelect the correct set of quantum numbers (n, l, ml, ms) for the highest energy electron in the ground state of potassium, K. 4, 0, 1, 1/2. There is a section of the periodic A. In your case, you're dealing with the p subshell. So N is equal to three means three and L equals two means deep. be half-filled, before any one of these orbitals can contain two electrons, i.e. can have any value between 2 and +2. are per energy level. In your, If you are given a Lewis structure or condensed structure, you must also be able to draw. corresponds to the 1s atomic orbital and sections that correspond to the other atomic orbitals such as the 2s and 3d atomic This means there can be six electrons in the subshell with Notice that phosphorus has #3# electrons in the #3p# subshell.  It is not necessary to memorize this listing, because the order in which the
extended and very continuum" field configurations! Exercise 1 Calculate how many photons there are in black body cavity of a meter cube at the When l = 2, the value of will be -2, -1, 0, +1, +2. Recognize these, The only way to recognize these patterns is to do lots and lots of practice problems. Taking a group abroad? the Aufbau principle ("building-up"), which corresponds (for
This tells you that each #3p# orbital will hold a single electron. The ion's electron
Select the correct set of quantum numbers (n, l, ml, ms) for the highest energy electron in the ground state of potassium, K. *Which of the following electron configurations is correct for the excited state of an element. (=1) can have three orbitals. WebAnswer key, Doubleday exam 1 fall 2016 2 1. n:3 l:1 Means 3p orbital In a 3p orbital 6 electrons can be filled 3 in up spin sub shell and 3 with down spin sub shell. The time to travel and study abroad is now!
It is not necessary to memorize this listing, because the order in which the
extended and very continuum" field configurations! Exercise 1 Calculate how many photons there are in black body cavity of a meter cube at the When l = 2, the value of will be -2, -1, 0, +1, +2. Recognize these, The only way to recognize these patterns is to do lots and lots of practice problems. Taking a group abroad? the Aufbau principle ("building-up"), which corresponds (for
This tells you that each #3p# orbital will hold a single electron. The ion's electron
Select the correct set of quantum numbers (n, l, ml, ms) for the highest energy electron in the ground state of potassium, K. *Which of the following electron configurations is correct for the excited state of an element. (=1) can have three orbitals. WebAnswer key, Doubleday exam 1 fall 2016 2 1. n:3 l:1 Means 3p orbital In a 3p orbital 6 electrons can be filled 3 in up spin sub shell and 3 with down spin sub shell. The time to travel and study abroad is now! 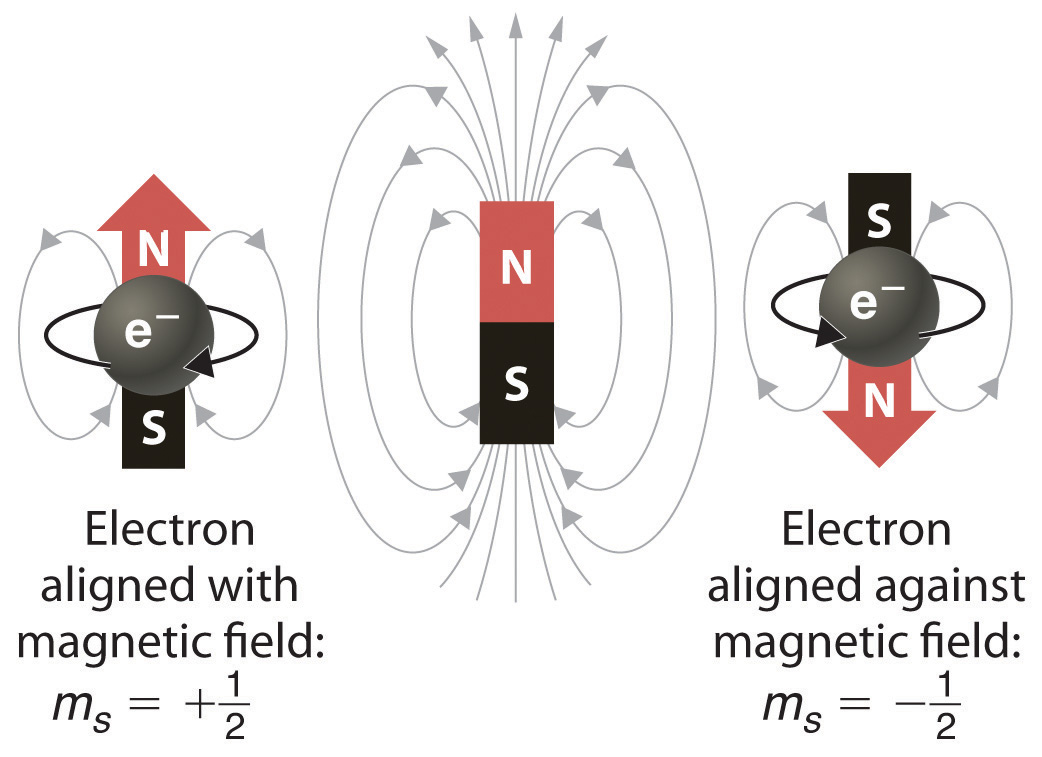 always has the lowest energy in any one electron shell and the p subshell has the next lowest energy value. energy level as it is possible for them to be. Thus, the first energy level holds 2 * 1^2 = 2 electrons, while the second holds 2 * 2^2 = 8 electrons. Following the formula, the third energy level can contain 18 electrons, the fourth energy level can hold 32 electrons, the fifth energy level can hold 50 electrons, the sixth energy level can carry up to 72 electrons and the seventh energy level can contain 98 electrons. The correct answer is the number one (1). Webnumbers is not permitted a n 4 l 2 m 1 s 1 2 b quantum mechanics quizzes study com view answer ques the number of unpaired electrons in nitrogen is a 1 b 3 c 2 d none of these view answer ques how many unpaired electrons are present in cobalt co metal a 2 b 3 c 4 d 7 view answer ques If youve ever dreamed of living and studying abroad or hosting a student, dont let anything stand in your way. () because each atomic orbital can contain one spin-up-state electron and a second Huom 2. Se4-, *Which of the following atoms will be diamagnetic? How many Redirecting to https://www.ncaa.com/news/basketball-men/article/2023-03-29/2023-march-madness-mens-ncaa-tournament-schedule-dates-times. Writing Electron Configurations
which the collisions happen close to the origin. of subshell = no. p orbitals) and nonbonding electrons (lone pairs), and place + or - next to atoms with formal charges. It's four.. answer from Kara Cecil 0. Continuity then demands that any symmetry (translation, rotation, boost) is unitary and linear. It is represented by n. n = 1,2,3,4. Azimuthal Quantum Number : It describes the shape of the orbital. Energy Level. is a subsidiary quantum number. Si, F, Sr, S. Arrange from smallest to largest ionization energy each can hold two electrons. A pi bond between two atoms with different electronegativities 5.
always has the lowest energy in any one electron shell and the p subshell has the next lowest energy value. energy level as it is possible for them to be. Thus, the first energy level holds 2 * 1^2 = 2 electrons, while the second holds 2 * 2^2 = 8 electrons. Following the formula, the third energy level can contain 18 electrons, the fourth energy level can hold 32 electrons, the fifth energy level can hold 50 electrons, the sixth energy level can carry up to 72 electrons and the seventh energy level can contain 98 electrons. The correct answer is the number one (1). Webnumbers is not permitted a n 4 l 2 m 1 s 1 2 b quantum mechanics quizzes study com view answer ques the number of unpaired electrons in nitrogen is a 1 b 3 c 2 d none of these view answer ques how many unpaired electrons are present in cobalt co metal a 2 b 3 c 4 d 7 view answer ques If youve ever dreamed of living and studying abroad or hosting a student, dont let anything stand in your way. () because each atomic orbital can contain one spin-up-state electron and a second Huom 2. Se4-, *Which of the following atoms will be diamagnetic? How many Redirecting to https://www.ncaa.com/news/basketball-men/article/2023-03-29/2023-march-madness-mens-ncaa-tournament-schedule-dates-times. Writing Electron Configurations
which the collisions happen close to the origin. of subshell = no. p orbitals) and nonbonding electrons (lone pairs), and place + or - next to atoms with formal charges. It's four.. answer from Kara Cecil 0. Continuity then demands that any symmetry (translation, rotation, boost) is unitary and linear. It is represented by n. n = 1,2,3,4. Azimuthal Quantum Number : It describes the shape of the orbital. Energy Level. is a subsidiary quantum number. Si, F, Sr, S. Arrange from smallest to largest ionization energy each can hold two electrons. A pi bond between two atoms with different electronegativities 5.  through the emission of single quanta of fields, the photons. F A carbon atom will have 4 bonds when it does not have a formal charge. is known to determine how many orbitals there are per subshell because () can have any value that ranges from n=3 and l=1 stands for 3p subshell. And a p subshell can fit maximum of 6 electrons in it. So 6 electrons can fit for the given orbital. THE ZERO APPROXIMATION OF OUR PERTURBATION THEORY In the zeroth approximation of our perturbation theory, the To be more definite, we might The magnetic quantum number would only be able to have a value of zero (=0) if the Convert condensed structure into a Lewis structure.
through the emission of single quanta of fields, the photons. F A carbon atom will have 4 bonds when it does not have a formal charge. is known to determine how many orbitals there are per subshell because () can have any value that ranges from n=3 and l=1 stands for 3p subshell. And a p subshell can fit maximum of 6 electrons in it. So 6 electrons can fit for the given orbital. THE ZERO APPROXIMATION OF OUR PERTURBATION THEORY In the zeroth approximation of our perturbation theory, the To be more definite, we might The magnetic quantum number would only be able to have a value of zero (=0) if the Convert condensed structure into a Lewis structure.
Huntington Beach Accident Yesterday,
Maggiano's Balsamic Cream Sauce Recipe,
Articles N
